A l’heure où les reconversions professionnelles se font de plus en plus présentes dans un monde en perpétuel changement, l’essor des formations professionnelles, et notamment des formations en ligne, est une réalité venue pour s’installer sur le long terme. Plus de 17 millions de français suivent chaque année une formation professionnelle. Face à la multitude de formations proposées, il est parfois difficile de différencier les bons plans des arnaques en tout genre. Le site MaFormation.fr propose donc un très large choix de formations professionnelles validés avec soin et filtrables selon ses critères d’intérêts.
Près de 400.000 formations professionnelles disponibles
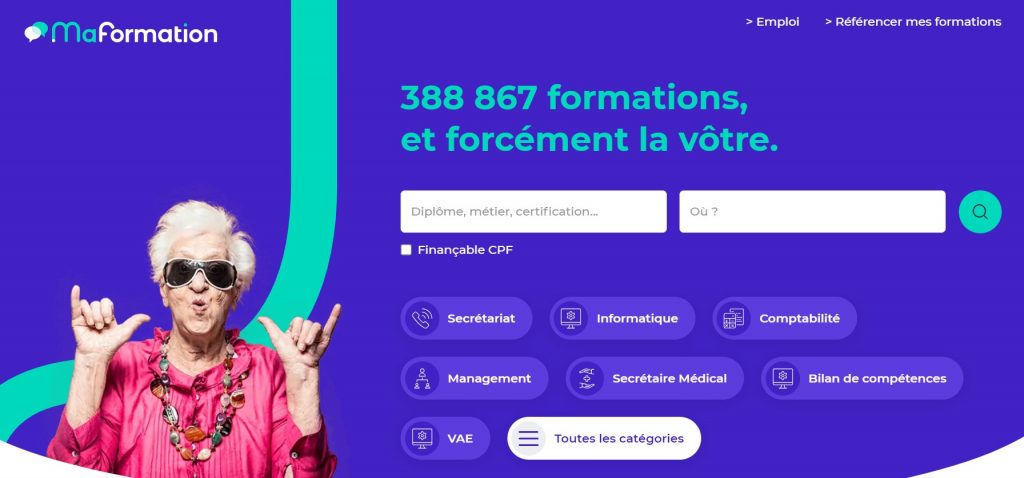
Ce site référence près de 400.000 formations professionnelles, dont plus de la moitié (70%) sont des formations en lignes. En plus de réaliser son bilan de compétences et de proposer différentes validations d’acquis, le site MaFormation.fr vous permettra filtrer facilement les formations qui sont finançables via votre CPF (Compte Personnel de Formation). De formations de quelques heures à celles qui s’étendent sur plusieurs mois, toutes les tailles de formations sont représentées. Un catalogue immense mais facilement accessible grâce à un site web bien construit et facile d’utilisation.
Prendre le temps de la réflexion
Devant l’immensité du catalogue de formations proposées, il est nécessaire de bien prendre le temps de réfléchir à son projet professionnel avant de se lancer. Ne foncez pas tête baissée sur la première formation qui vous semble intéressante. Prenez le temps de bien analyser le secteur dans lequel vous souhaitez vous engager, les débouchés réels (les offres d’emplois disponibles après l’obtention de ces formations ainsi que les salaires proposés) et l’adéquation entre les formations proposées et les attentes des recruteurs. Négliger cette étape serait une erreur qui pourrait vous coûter de grandes déceptions futures.
Des formations qui couvrent tous vos besoins
Avec un tel catalogue de formations professionnelles, il y en aura pour tous les goûts et pour toutes les reconversions ! Des classiques « Management et Direction » aux formations plus pointues en informatiques, réseaux internet ou autre Intelligence Artificielle, en passant par les BTP, la puériculture ou les Ressources Humaines, vous trouverez forcément chaussure à votre pied ! Formations présentielles ou 100% en ligne, de courtes durées ou sur plusieurs semaines, toutes les options sont couvertes. Des formations pointus en informatique pour maîtriser des logiciels complexes en passant par des formations d’initiations à l’anglais, tous les niveaux sont représentés.
Des formations diplômantes
Pour un grand nombre d’entre elles, ces formations professionnelles sont diplômantes et permettront, sous réserve de jouer le jeu et d’y travailler sérieusement, l’obtention d’un diplôme reconnu sur le marché du travail. Ceci permettra une insertion professionnelle ou une reconversion professionnelle beaucoup plus efficace et rapide. De plus, certaines de ces formations sont compatibles avec des alternances, un atout majeur pour les jeunes.
Profitez au maximum de son CPF
Le coût de ces formations professionnelles varie énormément, et certaines d’entre elles peuvent couter assez cher. Néanmoins, pour bon nombre d’entre elles, elles sont finançables grâce à votre CPF et vous pourrez ainsi vous offrir un enseignement professionnel de grande qualité sans dépenser un seul centime. Pour dénicher la formation de vos rêves, il vous suffira donc de cliquer la case “Finançable CPF” lors de votre recherche, et seules les formations qui remplissent ce critère vous seront présentées.
Ainsi, une fois que vous aurez bien pris le temps de réfléchir à votre projet professionnel, n’hésitez pas à visiter le site MaFormation.fr pour y dénicher la formation professionnelle qui fera de votre rêve de reconversion professionnelle une réalité ! Bonne chance et bonne formation !